|
5/7/2023 0 Comments Keynote 522 
The study did not report any new safety issues. The safety profile was similar to chemotherapy and Keytruda monotherapy in patients with high-risk early TNBC. 76.8 percent in the control group.Īt the median follow-up of 15.5 months, the Keytruda group’s PCR was 64.8 percent, much higher than 51.2 percent of the control group. The EFS in the third year was 84.5 percent in the treatment group vs. 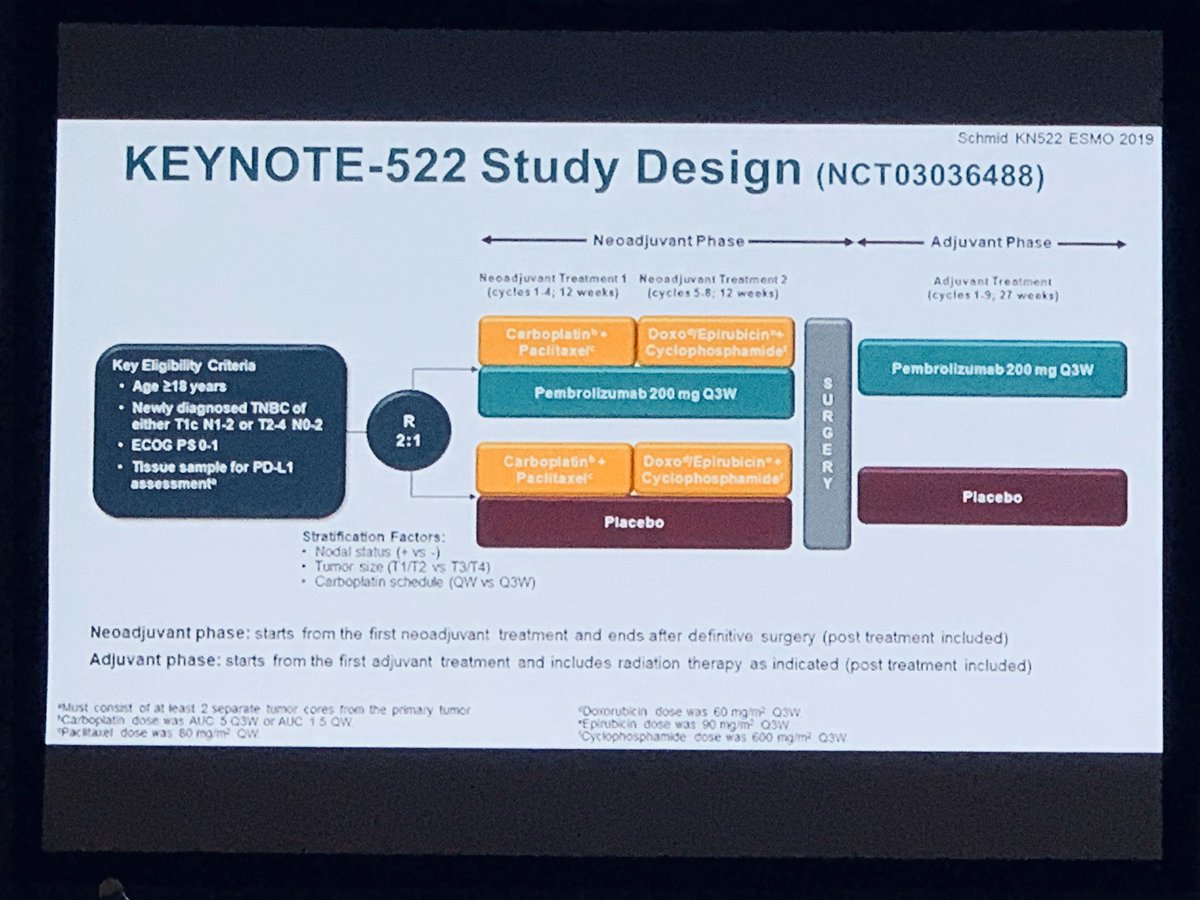
The study also demonstrated that Keytruda prolonged the EFS significantly. The study outcome showed that the Keytruda-chemotherapy combination group had a 37 percent lower risk of disease progression that precluded definitive surgery, a local or distance recurrence, a second primary cancer, or death from any cause compared to the chemotherapy alone group. The latest nod was based on the phase 3 KEYNOTE-522 trial in 1,174 TNBC patients. Along with postoperative adjuvant therapy, it aims to prevent metastasis and recurrence. Usually, neoadjuvant therapy before surgery aims to raise complete pathological response (pCR) in the short term to increase event-free survival (EFS) and overall survival (OS). 
To raise survival and prevent relapse in TNBC, MSD set pre-operative and post-operative adjuvant therapy as “one treatment strategy.” The latest indication allows Keytruda to treat both early and metastatic TNBC. In July last year, Keytruda obtained the permit as combination therapy with chemotherapy to treat locally recurrent unresectable or metastatic TNBC with positive PD-L1 expression. The indication does not require a PD-L1 expression test, giving benefits to more breast cancer patients.Īlso, the government approved using Keytruda every three weeks and every six weeks before or after surgery, which is expected to improve patient convenience. 
It continued as monotherapy after surgery in stage 2 and stage 3 TNBC patients who have not received prior treatment. On Wednesday, the Ministry of Food and Drug Safety authorized Keytruda as neoadjuvant therapy in combination with chemotherapy (carboplatin and paclitaxel, followed by doxorubicin or epirubicin and cyclophosphamide) before surgery. Last month, Merck and AstraZeneca reported that Lynparza mitigated the risk of invasive recurrences in patients with germline BRCA-mutated high-risk human epidermal growth factor receptor 2 negative early breast cancer in the Phase III OlympiA trial.MSD’s anti-PD-1 therapy Keytuda obtained expanded indication to be neoadjuvant therapy in combination with chemotherapy before surgery in a patient with triple-negative breast cancer (TNBC) and then continued as a single agent after surgery. Merck has submitted the latest results from KEYNOTE522 to the US Food and Drug Administration and is working with the regulator on the review of its application. “KEYNOTE-522 is the first large, randomised Phase III study to report a statistically significant and clinically meaningful EFS result among patients with stage II and stage III TNBC.” Merck Research Laboratories clinical research vice-president Dr Vicki Goodman said: “These highly-anticipated event-free survival results in this TNBC population build upon earlier findings from the KEYNOTE-522 trial and further support the potential use of Keytruda in these patients. The safety of the Keytruda combination was similar to the known profiles of individual regimens, without any new safety concerns. The trial is progressing for further follow-up of a key secondary goal, overall survival.ĭuring this fourth interim analysis, Keytruda offered a 28% decrease in the risk of mortality compared with a chemotherapy-placebo regimen. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
